Neutron activation analysis (NAA)
For chemical analysis
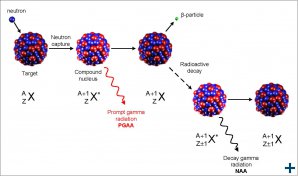
Neutron Activation Analysis (NAA) is a nuclear analytical technique. After the capture of a neutron by the atomic nucleus in a sample, gamma rays with an isotope specific energy are emitted. Their detection can be used for non-destructive, highly sensitive isotope analysis of a large variety of materials.
Advantages of NAA
- Minimal sample handling prior to irradiation leading to a minimal risk of contamination
- Possibility to perform analysis non-destructively, using INAA
- Large dynamic range of concentrations, which can be determined, from μg kg-1 to tens of percent
- Large dynamic range of sample masses, from a few mg to several hundred mg
- High trueness and low uncertainty of results, which are traceable to the SI unit of mass fraction, because NAA with relative standardization has recently been recognized as a primary ratio method
- Both relative and k0-NAA standardization available, the latter especially suitable for panoramic analyses
- NAA procedures with radiochemical separation (radiochemical neutron activation analysis – RNAA) available for determination of selected elements, namely V, Cr, Mn, Co, Ni, As, Se, Mo, Ag, Cd, Sb, I, rare earth elements, Re, Pt, Au, and Hg.
- Quality control achieved through the use of certified reference materials and participation in international proficiency testing.
Typical applications
- Environmental control and monitoring – analysis of aerosols, soils, sewage sludges, biomonitors
- Geo- and cosmochemistry – elemental characterization of rocks, minerals, tektites, meteorites
- Geomycology – elemental characterization of mushrooms and their substrates
- Nutritional science – determination of essential and toxic trace elements in foodstuffs and beverages
- Biomedicine – determination of essential and toxic trace elements in human and animal tissues
- Materials science – elemental characterization of various materials, e.g. high-tech materials that are difficult to analyse by other analytical methods
- Chemometry – quality control analyses to check performance of other analytical techniques, homogeneity testing and certification analyses of newly prepared reference materials
Further Information/Source: http://canam.ujf.cas.cz/en/home/item/176